
Ethanol(alcohol)---CH3CH2OH An alcohol is an organic compound that contains the hydroxyl group (-OH). Alcohols are class classified according to number of other carbon atoms that are directly bonded to carbon atom attached to hydroxyl group. the picture shows what is alcohol.
Reference:http://en.wikipedia.org/wiki/Alcohol
Unit 1 organic chemistry
Reference:http://en.wikipedia.org/wiki/Alcohol
Unit 1 organic chemistry

Hydroxyl group (-OH) A hydroxyl group is a functional group that an oxygen atom connected by a hydrogen atom, we can see it as a substructure of the water molecule. When it appears, it has a properties of reactive and interactive. -OH looks like the hydroxide ion of water (OH-, associated with ionizability)The neutral form of the hydroxyl group is a hydroxyl radical.
Reference:http://en.wikipedia.org/wiki/Hydroxyl
Unit 1 organic chemistry
Reference:http://en.wikipedia.org/wiki/Hydroxyl
Unit 1 organic chemistry
Reaction involving alcohol: Dehydration reaction, Esterification, Oxidation

Dehydration reaction

Oxidation reaction

Esterification reaction
Properties of alcohols: hydrogen bonding
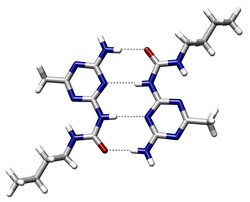
A strong dipole-dipole force between a hydrogen atom attached to a highly electron-negative atom(N,O,or F) in one molecule and a highly electron-negative atom in another molecule.
Alcohols typically have much higher boiling points than the corresponding substituted alkanes. For example, the boiling point of methanol is 65℃ while the boiling point of methane is -162℃.
reference: text book intermolecular forces
unit 4 chemical bonding
Alcohols typically have much higher boiling points than the corresponding substituted alkanes. For example, the boiling point of methanol is 65℃ while the boiling point of methane is -162℃.
reference: text book intermolecular forces
unit 4 chemical bonding
单击此处进行编辑。
