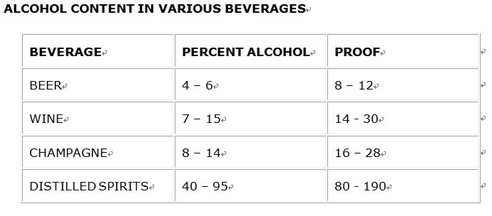
There are differences in the amount of alcohol between beer, wine, champagne and distilled spirits. The amount of alcohol is given as a percentage and also in "proof". The proof of an alcohol beverage is equal to twice the percentage of ethyl alcohol.
reference:http://www.chemcases.com/alcohol/alc-03.htm

As a fuel
Ethanol burns to give carbon dioxide and water and can be used as a fuel in its own right, or in mixtures with petrol (gasoline). "Gasohol" is a petrol / ethanol mixture containing about 10 - 20% ethanol. Because ethanol can be produced by fermentation, this is a useful way for countries without an oil industry to reduce imports of petrol.
C2H5OH + 3O2 --> 2CO2 + 3H2O
The standard enthalpy of combustion of ethanol is -1368 kJ /mol.
the standard enthalpy change of combustion of ethanol is -1370.7 kJ/mol.
the standard enthalpy of formation of ethanol is -276.5 kJ/mol.
reference:http://www.chemcases.com/alcohol/alc-03.htm
Ethanol burns to give carbon dioxide and water and can be used as a fuel in its own right, or in mixtures with petrol (gasoline). "Gasohol" is a petrol / ethanol mixture containing about 10 - 20% ethanol. Because ethanol can be produced by fermentation, this is a useful way for countries without an oil industry to reduce imports of petrol.
C2H5OH + 3O2 --> 2CO2 + 3H2O
The standard enthalpy of combustion of ethanol is -1368 kJ /mol.
the standard enthalpy change of combustion of ethanol is -1370.7 kJ/mol.
the standard enthalpy of formation of ethanol is -276.5 kJ/mol.
reference:http://www.chemcases.com/alcohol/alc-03.htm

Antifreeze:
A 50% v/v (by volume) solution of ethylene glycol in water is commonly used as an antifreeze.
reference:http://en.wikipedia.org/wiki/Alcohol#Applications
A 50% v/v (by volume) solution of ethylene glycol in water is commonly used as an antifreeze.
reference:http://en.wikipedia.org/wiki/Alcohol#Applications

Antiseptics:
Ethanol can be used as an antiseptic to disinfect the skin before injections are given, often along with iodine. Ethanol-based soaps are becoming common in restaurants and are convenient because they do not require drying due to the volatility of the compound. Alcohol based gels have become common as hand sanitizes.
reference:http://en.wikipedia.org/wiki/Alcohol#Applications
Ethanol can be used as an antiseptic to disinfect the skin before injections are given, often along with iodine. Ethanol-based soaps are becoming common in restaurants and are convenient because they do not require drying due to the volatility of the compound. Alcohol based gels have become common as hand sanitizes.
reference:http://en.wikipedia.org/wiki/Alcohol#Applications

Solvents:
hydroxyl groups (-OH), found in alcohols, are polar and therefore hydrophilic (water loving) but their carbon chain portion is non-polar which make them hydrophobic. The molecule increasingly becomes overall more nonpolar and therefore less soluble in the polar water as the carbon chain becomes longer. Methanol has the shortest carbon chain of all alcohols (one carbon atom) followed by ethanol (two carbon atoms.)
reference:http://en.wikipedia.org/wiki/Alcohol#Applications
hydroxyl groups (-OH), found in alcohols, are polar and therefore hydrophilic (water loving) but their carbon chain portion is non-polar which make them hydrophobic. The molecule increasingly becomes overall more nonpolar and therefore less soluble in the polar water as the carbon chain becomes longer. Methanol has the shortest carbon chain of all alcohols (one carbon atom) followed by ethanol (two carbon atoms.)
reference:http://en.wikipedia.org/wiki/Alcohol#Applications
Unit 3 energy changes and rates of reaction
